-1.png)
“In preclinical studies, SBT6050 demonstrated a broad therapeutic window and this profile has enabled the selection of a first-in-human starting dose projected to be pharmacologically active,” Odegard added, “SBT6050 is designed for systemic administration and tumor-localized activation of myeloid cells, in order to circumvent toxicities associated with untargeted myeloid cell agonists,” said Valerie Odegard, Ph.D., Silverback’s chief scientific officer. The approach of myeloid cell activation and reprogramming in the tumor microenvironment has emerged as a promising therapeutic approach. The investigational agent is designed to activate human myeloid cells in tumors expressing moderate or high levels of HER2. SBT6050, a novel ImmunoTAC™ therapeutic being developed by Silverback Therapeutics, includes a HER2-directed antibody conjugated to a potent and specific small molecule toll-like receptor 8 (TLR8) agonist, with activity localized to HER2-expressing tumor sites. TLR8 is highly expressed in myeloid cells that are prevalent in human tumors and TLR8 agonism can potently activate a broad spectrum of anti-tumor immune mechanisms. Īctivation of toll-like receptor 8 (TLR8) enhances natural killer(NK-) cell activation, increases antibody-dependent cell-mediated cytotoxicity and induces Th1 polarizing cytokines. TLR-dependent recognition of PAMPs leads to activation of the innate immune system, which subsequently leads to the activation of antigen-specific adaptive immunity. TLRs recognize specific patterns of microbial components, called pathogen-associated molecular patterns (PAMPs). Valerie Odegard, Ph.D., has been named president and chief scientific officer, and Naomi Hunder, M.D., has been named chief medical officer.The innate immune system recognizes invading microorganisms by a phylogenetically conserved family of proteins PRRs of which toll-like receptors (TLRs) are one of the most important. The company also announced two key executive promotions. 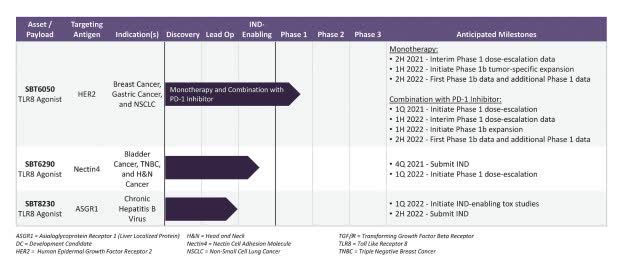
SBT6050 is currently in a Phase 1 clinical study in patients with advanced or metastatic HER2-expressing solid tumors ( Identifier: NCT04460456). Silverback’s lead product candidate, SBT6050, is a therapeutic comprised of a TLR8 agonist payload conjugated to a HER2-directed monoclonal antibody that targets tumors such as certain breast, gastric and non-small cell lung cancers. Initially, the company is creating a new class of targeted immuno-oncology agents that direct a TLR8 agonist myeloid cell activator to the tumor microenvironment in solid tumors to promote cancer cell killing. Its platform enables the strategic pairing of proprietary payloads that modulate key disease modifying pathways with monoclonal antibodies directed at specific disease sites. Silverback Therapeutics is a clinical-stage biopharmaceutical company focused on leveraging its proprietary ImmunoTAC TM technology platform to develop systemically delivered and tissue targeted therapeutics for the treatment of cancer and other serious diseases. The company intends to use the proceeds to support its clinical development of SBT6050, a TLR8 agonist conjugated to a HER2-directed antibody currently in a Phase 1 clinical study for the treatment of HER2-expressing solid tumors, and to advance its pipeline of other ImmunoTAC therapeutics. Venture Partners, Nextech Invest Ltd., Hunt Technology Ventures, and Pontifax Venture Capital. The round was led by EcoR1 Capital with participation from new investors including Boxer Capital of Tavistock Group, Fidelity Management & Research Company LLC, Nantahala Capital Management, and RA Capital and existing investors OrbiMed Advisors, U.S. Silverback Therapeutics, Inc., a Seattle, WA-based clinical-stage biopharmaceutical company leveraging its proprietary ImmunoTAC technology platform to develop systemically delivered and tissue-targeted therapeutics for the treatment of cancer and other serious diseases, closed an $85m Series C financing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
